INTRODUCTION
Bisphosphonates (BP) are a group of drugs used to treat metastatic malignancies related to breast, prostate and other bone diseases such as osteoporosis and Paget’s disease. Its mechanism of action reduces bone resorption, inhibition of recruitment and promotion of osteoclast apoptosis. Despite the great benefits for patients under these conditions, a complication associated with its use is jaw osteonecrosis.
Patients with osteoporosis, cancer and those with metastatic bone disease often have complications including pain, pathological fracture, spinal cord compression and hypercalcemia, which worsen life quality, increasing morbidity and mortality.1 These changes are usually a consequence of the metastatic process, which is the result of osteoclast activation, mediated by different cytokines produced by tumor cells, which cause bone resorption allowing tumor growth.2
To control these complications, recently medicine has been using drugs called bisphosphonates (BPs), which are analogues of pyrophosphates, not metabolized, able to deposit in bone and inhibit osteoclastic function. These drugs are part of the treatment protocol for patients with moderate to severe cancer-associated hypercalcemia; patients with osteolytic lesions associated with breast cancer and multiple myeloma in combination with antineoplastic chemotherapy and for osteolytic lesions originating from any solid tumor.3,4 This has resulted in widespread use of bisphosphonates in most medical oncology clinics and hospitals worldwide. Several clinical studies have shown the efficacy of BF’s in reducing osteoclast-mediated bone lysis, bone pain reduction and skeletal complications.1,5–10
From 2003, several case reports began to be published in the literature regarding a serious complication possibly induced by bisphosphonate class drugs: bisphosphonate-induced jaw osteonecrosis. The American Association of Oral and Maxillofacial Surgeons has defined this condition as: “Bone tissue exposed in the maxillary facial region that persists for more than eight weeks in patients undergoing current or previous treatment with bisphosphonates, who have no history of head and neck radiotherapy.”
LITERATURE REVIEW
The bisphosphonate is a structural analogue of pyrophosphate, the product normal of human metabolism that, when it undergoes some structural modifications, gives rise to different generations of bisphosphonates with distinct activity levels.8 The first generation includes etidronate, the second represents the amino bisphosphonates, such as alendronate and pamidronate, the third generation has a cyclic chain and its representatives are risedronate and zoledronate.7
Anti-resorptive properties of bisphosphonates increase approximately tenfold between each generation of the drug.7 Bisphosphonates reduce bone resorption in a dose-dependent manner, mainly by inhibiting recruitment and promoting osteoclast apoptosis, in addition to stimulating osteoblastic activity.11
The BP´s basically have two categories of chemical structure of the R2 chain, which are nitrogenous and non-nitrogenous BF´s. Both are internalized by osteoclasts in the process of bone resorption leading to cell death by apoptosis by different mechanisms of action. Non-nitrogenous BP’s, when metabolized by osteoclasts, become substrates for the synthesis of cytotoxic adenosine triphosphate (ATP) analogues that cause cell death. However, nitrogenous BFs, after reabsorption by osteoclasts seem to act by interrupting the mevalonate pathway, which is responsible for guiding cholesterol synthesis. The interruption of this mechanism causes intra-cellular vesicular transport to be compromised, causing cell death, and directly affecting bone resorption.12–23
BP’s can be given orally and intravenously and are well distributed in plasma and partly (about 50 %) absorbed by bone and the remainder is excreted through the kidneys without any modification. BF’s accumulate for long periods within the bone matrix, depending on the treatment duration and type of BF’s prescribed, so the drug may remain in the body for several years.24 The BP´s marketed in the Brazil differ in terms of their presentation, trademarks and indications, as shown in Figure 1.
Diagnostics, Clinical Characteristics and Treatments
The diagnosis is basically made through anamnesis and the clinical examination of the patient. Several signs and symptoms precede their clinical manifestations, highlighting pain, tooth mobility, mucosal edema, erythema, ulceration and when involving the maxilla, there is chronic sinusitis. It may occur spontaneously or in a region prior to dental surgery.
Bisphosphonate-associated osteonecrosis (BAO) may be asymptomatic for weeks, months, and years, but may result in pain or exposure of the mandibular or maxillary bone when located near ulcerated or infected lesions.25,26 Ancillary examinations may be requested for diagnostic clarification such as by panoramic radiography. The radiographic aspect of the BAO may be defined as diffuse bone sclerosis, presence of bone sequestration, periosteal reaction and an antral fistula, as well as diffuse radiolucent patches and osteolytic lesions with involvement of cortical bone.27
CT scans show more detailed images that may help in the differential diagnosis between jaw osteonecrosis and metastatic bone disease. There may be resorption of the trabecular bone with alteration of its structure, which will depend greatly on the size and intensity of the BAO lesion in process.28
Another test that can be used, but with a preventive character, is the type I collagen type carboxyterminal telopeptide (CTX) test, which is recommended to assess the risk of osteonecrosis in those patients using bisphosphonates for more than three years. Levels greater than 150 pg/mL are ideal to perform any type of operation with minimal risk and without the need to discontinue medication. When less than 150 pg/mL, the medication should be stopped for a period of between four and six months or another prosthetic treatment option should be preferable.29
According to Assael’s publication (2009), the BAO levels are classified according to their respective stages, illustrated in Figure 2.
According to Souza et al from 2009, as published in their review, the association between the bisphosphonate and osteonecrosis is conclusive. However, the treatment protocols are still widely discussed as they involve systemic antibiotic therapy, debridement and/or resection of the tissues involved, hyperbaric therapy and/or microvascular surgery. See Figure 3 recreated from Souza et al 2009.
CASE REPORT
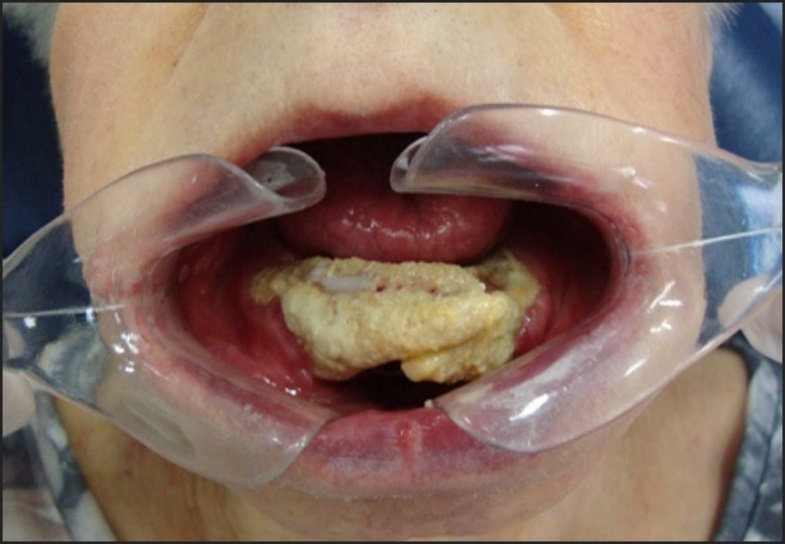
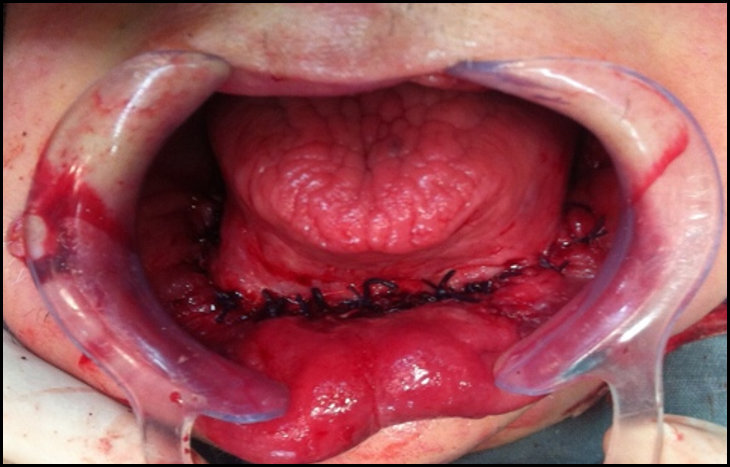
A 75-year-old female patient sought dental care reporting pain and discomfort in the mandibular region and difficulty in eating, with upper and lower total prosthesis and strong halitosis. There were areas of necrotic bone tissue exposed throughout the lower alveolar ridge associated with complete symphysis fracture. See Figure 4. The patient reported a history of previous breast cancer, surgical treatment associated with radio-therapy and intravenous chemotherapy, including Zometa for 6 years.
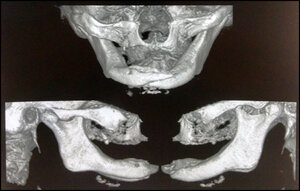
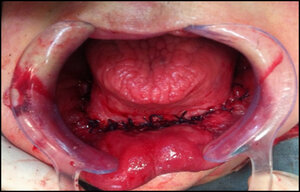
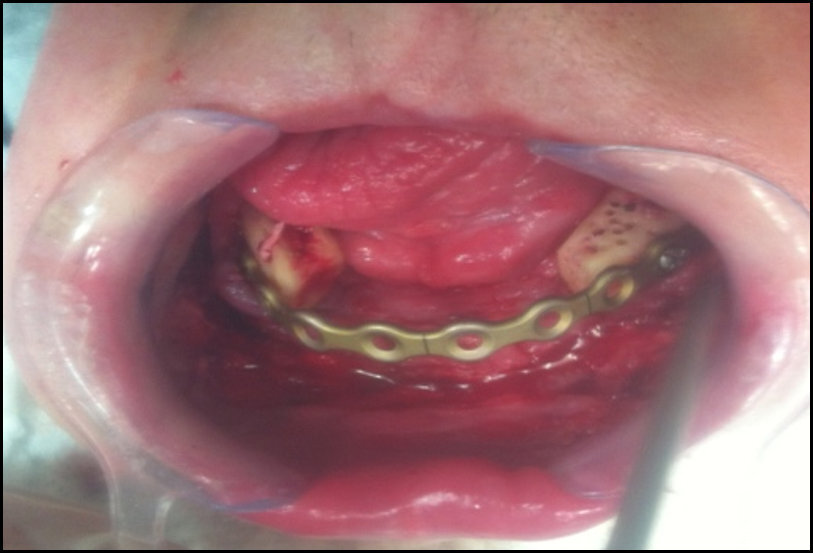
Radiographic exam revealed an extensive osteolytic lesion in the alveolar ridge and fracture in the symphysis region with dislocation of the stumps. See Figures 5 & 6. The proposed treatment was debridement of necrotic bone plus resection of stumps with safety margin, installation of reconstructive plate previously molded in a prototyping model (Figures 7, 8 & 9), the procedure was performed in a hospital environment under general anesthesia.
DISCUSSION
According to the American Association of Oral and Maxillofacial Surgery, the BAO must meet the following inclusion criteria17:
-
Previous or current treatment with bisphosphonates.
-
Necrotic bone exposed in the maxillo-facial region, which persists for more than eight weeks.
-
No previous history of radiotherapy in the jaw region.
Bisphosphonate-induced osteonecrosis lesions usually appear as mucosal ulcerations with underlying bone exposure and associated pain, although one-third are quite painless.18 The predominant location is in the mandible, although there are also reports of cases in the maxilla and palate.19 This preferential location seems to be related to the anatomical and physiological characteristics of the mandible, specifically, its lesser vascularization, as well as the terminal character of the mandibular artery. These lesions do not heal over a period of six to eight weeks, becoming persistent and not reacting to conventional treatments.20 The most common triggering factors for BAO are extraction and iatrogenic trauma, although they may also occur spontaneously.20
The inability of the hypodynamic and hypo-vascularized bone to compensate for any need for bone repair and remodeling due to physiological stress (chewing), iatrogenic trauma such as a poorly adapted prosthesis, surgical procedures or dental infections has contributed to the appearance of injuries.21,22 Predisposing factors, such as the use of drugs with anti-angiogenic properties (glucocorticoids, thalidomide, etc.) or diabetes mellitus and peripheral vascular disease, are all systemic risk factors.22
BAO caused by orally administered drugs differs significantly when associated with the intra-venous form in three main forms: patients taking oral bisphosphonates require a long period of therapy before evidence of exposed bone; manifest less bone exposure and symptoms are less severe and have a chance to improve symptoms or exposure after drug withdrawal, leading to bone healing.23
CONCLUSIONS
Indeed, given the very serious complications associated with the maxillofacial complex, all bisphosphonate treatment, particularly in patients with multiple myeloma and solid tumors, must be preceded by consultation with a dental surgeon with a view to adopting preventive measures, in particular:
-
Frequent consultation with the dental surgeon for oral condition assessment, hygiene control, fluoride application, radiographic monitoring, fitting of prostheses (every six months);
-
Stabilization of oral cavity diseases;
-
Avoid surgical acts in the oral cavity (extraction, implant placement, etc., after starting bisphosphonate therapy);
-
When invasive mouth procedures are required, the case should be discussed between the oncologist and the dentist.
-
Monitoring of bone tissue through Plasma CTX level.30
-
Clarification of the patient regarding risk factors for the development of osteonecrosis.
Such measures will allow the professional a more accurate analysis of the case, in order to reduce possible risks of lesion development or of any lesion reaching the later stages. Also, by utilizing the methodology of a multidisciplinary approach to patients, the clinician can help to select the optimal type of treatment for each patient, improve the prognosis and the quality of life.