INTRODUCTION
Myofibromas are uncommon benign myofibroblastic tumors in all tissues of the body, rarely found in the oral cavity. In 1989, a solitary case of this tumor was described by Smith et al. and called “myofibroma”.1 Subsequently, the terms “myofibromatosis” and “myofibroma” were adopted by the World Health Organization. idiopathic cause that presents a wide differential diagnosis, being clinically confused with more frequent and routine pathological entities in stomatology, among which we can mention: Pyogenic Granuloma, Peripheral Lesion of Giant Cells, Traumatic Fibroma and Peripheral Ossifying Fibroma2,3
OBJECTIVE
The purpose of this study is to report a case of myofibroma in the palatine gingiva in the posterior region of the right maxilla and to describe its clinical, histopathological and immunohistochemical characteristics.
CASE PRESENTATION
A 40-year-old female patient attended the outpatient clinic within the Department of Stomatology and Buccomaxillofacial Surgery of the Sorriso Institute, complaining of an increase in volume in the gingiva with an evolution time of approximately 5 months.
In the anamnesis, no systemic disorders, a non-smoker patient and a good general health status were reported. It was presented with blood pressure of 120 x 80 mmHg and glycemia 90 mg/dL.
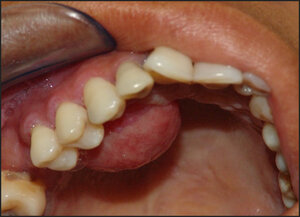
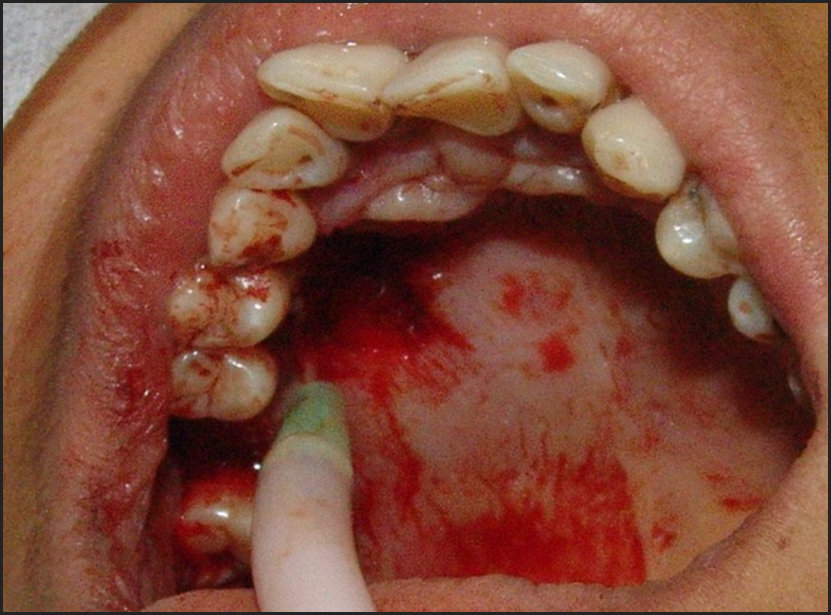
At the clinical examination, we observed a nodular-aspect lesion approximately 2 cm in diameter, painless, located in the palatine inserted gingiva near the right upper premolars, presenting a base of pediculated implantation, consistent with palpation, painless, smooth, bleeding surface when inspected, a pale pink color similar to the adjacent tissue (Figures 1 and 2).
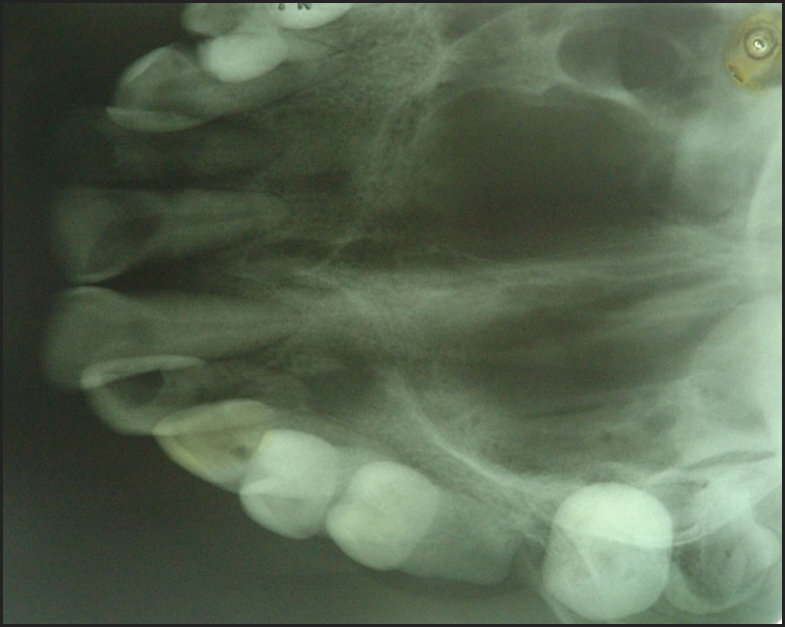
Based on anamnesis and clinical examination, we made the differential diagnosis of common lesions in the soft tissues of the oral cavity: Pyogenic Granuloma, Peripheral Lesion of Giant Cells, Traumatic Fibroma and Peripheral Ossifying Fibroma. Periapical and occlusal radiographic examinations were performed where we did not detect any type of bone alteration. (Figure 3)
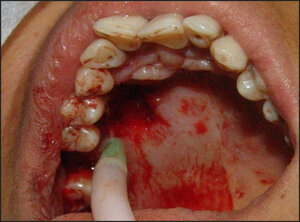
The proposed treatment was total excision of the lesion by incision with 15C scalpel blade, surrounding the entire lesion with safety margin performed under local infiltrative anesthesia of lidocaine with vasoconstrictor norepinephrine. The piece was wrapped in a vial containing 10% formalin and sent for anatomopathological examination. (Figure 4).
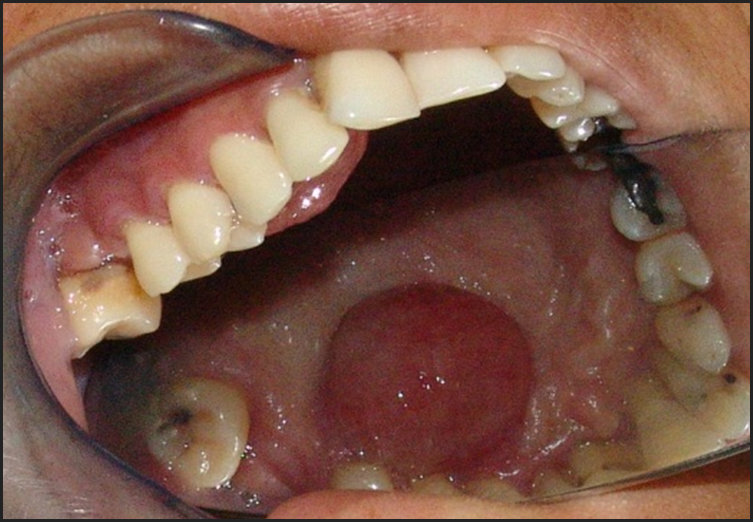
As it was a lesion in the palatine inserted gingiva and in the presence of the teeth in the region, it was not possible to coaptate the edges of the surgical wound by the conventional techniques of synthesis. We opted to cover the surgical site area with surgical cement based on zinc oxide and eugenol for the purpose of protection and favoring re-epithelialization. (Figure 5)
At the end of the surgery, antibiotic, non-steroidal anti-inflammatory and analgesic were prescribed, following the protocol: Amoxicillin 875mg, 12/12 hours for 7 days, Cetoprofeno 100mg, 8/8hours for 5 days and Paracetamol 750mg 6/6 hours for 4 days.
After 7 days the patient returned to the post-operative period where the surgical cement was removed and the aspect of the region was healing. The patient reported that there was no pain and edema in her post-operative recovery.
After one year, the patient returned to the Instituto do Sorriso to follow up on the case without symptomatology. The aspect of the region where Myofibroma was removed was healthy without signs of lesion recurrence.
Histopathological examination revealed fusiform cells that resemble fibroblasts or smooth muscle cells, suggesting myofibroblastic differentiations. See Figure 6. In the immunohistochemical examination, it showed positivity for HHF - 35 muscle-specific actin, vimentin, and alpha-smooth muscle actin (α-SMA) and with negativity for desmin, S-100 protein, CD34 and IHC. See Figure 7. Thus, it had the characteristics of a myofibromatosis lesion.
DISCUSSION & LITERAURE REVIEW
The term myofibromatosis was first used by Daimaru et al. in 1989, and later in the same year, a solitary case of this tumor was described by Smith et al. who called it myofibroma.1,2 Myofibroma and myofibromatosis are benign fibroblast tumors composed of myeloid cells with thin blood vessels surrounding the soft tissues, bones or internal organs. Solitary proliferation is called myofibroma and its multicentric manifestation is called myofibromatosis.1 The present case describes a solitary lesion of myofibroma in a female patient of 40 years old. Myofibroma is the most common fibrous proliferation in childhood, with a predisposition to the soft tissues of the head and neck.3 Myofibroma may appear from the newborn to old age,4 although 90% of these lesions appear before 2 years old.
Generally, myofibromas are twice as common in men as in women.3–5 The tongue is more commonly involved (30%), followed by the buccal mucosa (20%) and rarely seen in the gingiva, as in this case.3,6 In a recent study by Aiki et al., it was verified that, among 94 review cases, myofibromas involved the mandible (33%), gingiva (23%), tongue (15%), oral mucosa (12%), palate (8%), lip (4%) and other areas (5%), in that order.7
According to Vered et al., the histopathological characteristics of oral soft tissues myofibroma are in accordance with those described for the myofibroma of other parts of the body.8 However, an important difference was that while necrosis is a common finding in tumors involving other anatomical sites, only four cases of oral soft tissues myofibroma have demonstrated necrosis.8 It is suggested that areas of necrosis are associated with spontaneous regression of myofibroma.9 The occurrence of spontaneous regression of myofibromas may be mediated by apoptosis,9 which is seen in many normal embryonic and normal postnatal developmental processes. This may explain why the myofibromas that are present at birth or appear soon after, tend to regress spontaneously. It can be assumed that myofibroblasts that are influenced by various temporal and spatial factors become more resistant to apoptosis as the patient ages. Therefore, myofibroma lesions of oral soft tissues that usually develop in children or adults do not tend to regress spontaneously as in infants.
Histopathologically, myofibroma exhibits a biphasic pattern of light and dark areas. The appearance consists mainly of spindle cells with eosinophilic cytoplasm and tapered or cigar-shaped nuclei arranged in small fascicles or spirals and nodules at the periphery of the lesion. However, sometimes these cells are randomly distributed throughout the lesion. In contrast, the more intensely stained areas, more centrally located, consisted of round cells or small spindle cells arranged around thin-walled blood vessels similar to hemangiopericytoma with irregular branches. These cells have basophilic nuclei, small eosinophilic cytoplasm and undifferentiated cell margins.2,3,10 In some cases, light and dark areas are not separated, and the two subpopulations of cells are mixed.8 Mitotic figures are only rarely observed, but deeply localized lesions are often poorly defined and tend to focally infiltrate the surrounding tissue.10 Immunohistochemically, myofibroma cells express α-SMA, muscle specific actin and vimentin and are negative for desmin, S-100 protein and CD34.2,5,8,10–20 In our case, the histopathological, histochemical and immune-histochemical findings fulfill the criteria for the diagnosis of oral soft tissue myofibroma.
The histopathological differential diagnosis of soft tissue myofibroma of the mouth includes leiomyoma, schwannoma, benign fibrous histiocytoma, solitary fibrous tumor, desmoid fibromatosis.3,8 Neoplastic cells of vascular leiomyoma are positive for desmin, in contrast to negative myofibroma. In addition, Masson’s trichrome stain in vascular leiomyoma shows the presence of delicate fibrous tissue that surrounds smooth muscle cells and septa between the masses of neoplastic cells. This distribution is different from that of the thick fibrous bundles, with random angles, which intersect irregularly prominently seen in the myofibroma.3,11 Schwannoma and neurofibroma do not contain blood vessels of the hemangiopericytoma type and neoplastic cells are positive for the S-100 protein.8 Benign fibrous histiocytoma consists of fusiform cells resembling fibroblasts and histiocytic cells. These cells express factor XIIIa and α1-antitrypsin.3 Solitary fibrous tumor can be differentiated from myofibroma because its neoplastic cells express CD34.15 Unlike myofibroma, desmoid type fibromatosis is not characterized by biphasic cell populations.
The treatment for myofibroma is excisional biopsy. Local recurrence has been reported in 7% to 31% of cases,13 however, it is believed that these relapses are caused mainly by insufficient resection of the lesion. Chemotherapy or radiotherapy has been rarely used for the treatment of myofibromas and myofibromatosis, except for some cases of persistent recurrence or an unresectable lesion.14
CONCLUSIONS
Myofibroma is a rare non-neoplastic type of proliferative lesion of idiopathic etiology that can be clinically mistaken for other pathological entities of similar appearance that more frequently affect the oral cavity, among them we can mention Pyogenic Granuloma, Peripheral Lesion of Giant Cells, Traumatic Fibroma and Peripheral Ossifying Fibroma. It is a tumor that is difficult to diagnose, requiring immunohisto-chemical evaluation to complete the diagnosis. In the present case, the lesion was found in the gingiva inserted into the region of the upper premolars. The histopathological differential diagnosis of myofibroma in soft tissues of the mouth includes leiomyoma, schwannoma, benign fibrous histiocytoma, solitary fibrous tumor and desmoid fibromatosis. It is important for the dental surgeon to know the existence and possibilities of this type of lesion besides being prepared to make the diagnosis and propose the appropriate treatment.
Statement of Conflicts
None




.jpg)



.jpg)